(ANL), Argonne, IL (United States) Sponsoring Org. Publication Date: Research Org.: Ames Lab., Ames, IA (United States) Argonne National Lab. Department of Chemistry and Department of Chemical & Biological Engineering Northwestern Univ., Evanston, IL (United States).Department of Chemistry Argonne National Lab. Ar 3 s satellite spectrum studied by asymmetric ( e, 2e ) experiments Article Nov. and Iowa State Univ., Ames, IA (United States) Any procedure, which one is XPS peak fit software, Please any Researches. The binding energies of the palladium metal from the XPS outcomes are in great concurrence with reported values in the literature 43, 441). Department of Materials Science & Engineering The other pair of asymmetric peaks at 336.2, 341.9, and 343.4 eV attributed to Pd 3d3/2 signal from Pd-oxide species. Department of Chemical & Biological Engineering The well-defined nature of the single-site more » supported Pd2+ precursor provides direct mechanistic insights into this unusual and likely general reductive process. When these parameters take a value of 0, the peak is a pure Gaussian.
#Palladium asymmetric xps peak free
This process reasonably proceeds via an olefin activation/coordination/insertion pathway, followed by beta-hydride elimination to generate free Pd-0. The functions are calculated for a single peak with an FWHM of 400 channels and a peak maximum of 10 000 counts over a range of 4001 channels, 2000 channels to the right and left of the peak. Diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) and temperature-programmed reaction mass spectrometry (TPR-MS) reveal that the surface complex reduction with ethylene coproduces H-2, acetylene, and 1,3-butadiene. The binding energies of the palladium metal from the XPS outcomes are in great concurrence with reported values in the literature 43, 44 (Table 1). These nanoparticles are catalytically relevant using the aerobic 1-phenylethanol oxidation as a probe reaction, with rates comparable to a conventional Pd/Al2O3 catalyst but without an induction period. The other pair of asymmetric peaks at 336.2, 341.9, and 343.4 eV attributed to Pd 3d3/2 signal from Pd-oxide species. Indeed, upon exposure to gaseous ethylene or liquid 1-octene at 25 degrees C, the Pd2+ species is reduced to form Pd-0 nanoparticles with a mean diameter of 4.3 +/- 0.6 nm, as determined by scanning transmission electron microscopy (STEM). This surface complex is a precursor in the direct synthesis of size-controlled Pd nanoparticles under mild reductive conditions and in the absence of additional stabilizers or pretreatments.
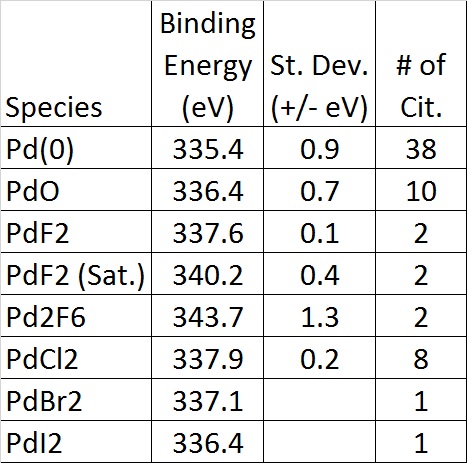
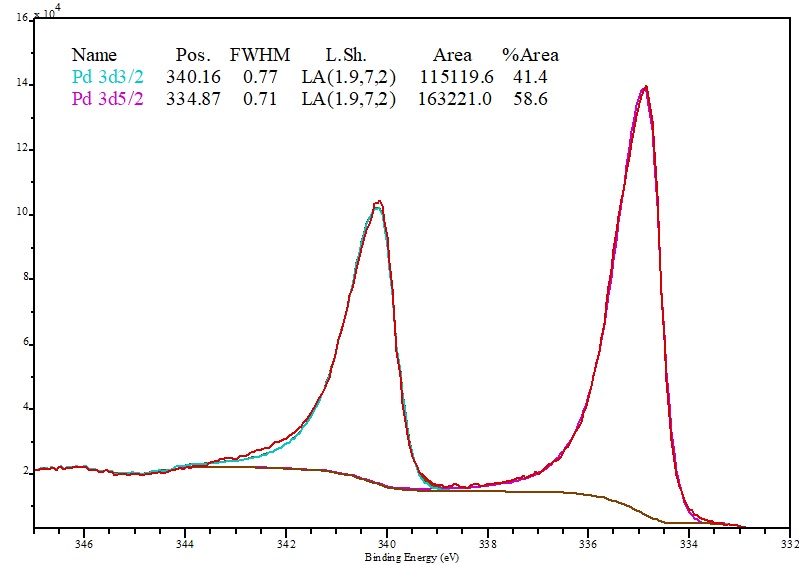
The resulting surface complex is characterized by inductively coupled plasma atomic emission spectroscopy (ICP-AES), X-ray photoelectron spectroscopy (XPS), X-ray absorption spectroscopy (XAS), and dynamic nuclear polarization surface-enhanced solid-state nuclear magnetic resonance spectroscopy (DNP SENS). The element can be found as a free metal and alloyed within platinum and gold in deposits located in Ethiopia, South and North America, and the Ural Mountains.A surface metal-organic complex, (-AlOx)Pd(acac) (acac = acetylacetonate), is prepared by chemically grafting the precursor Pd(acac)(2) onto gamma-Al2O3 in toluene at 25 degrees C. Palladium is commercially-produced from nickel-copper deposits. Palladium was named after the asteroid Pallas, which was discovered two years prior to the element’s discovery. 36 The Pd 3d peak at 335.8 eV corresponds to metallic palladium. which is likely to be a shake-up feature.
X-ray photoelectron spectra (XPS) were recorded on a Kratos XSAM 800 spectrometer. A very narrow asymmetric C 1 s peak at 284.5 eV was observed with minimal CO bonding for shortened empty GNF. Palladium was first discovered in 1803 in crude platinum ore from South America by W. Palladium nanoparticle-graphene catalysts for asymmetric hydrogenation.

When dissolved in hydrochloric acid, palladium absorbs up to 900 times its own volume of hydrogen at room temperature. asymmetric XPS signals with various peak shapes that are available to the XPS community, while also showing and emphasizing that the Voigt peak shape is a superior shape for peak fitting XPS data. Some notable features of palladium include its tarnish resistance, low density and melting point, and its chemical attraction to sulfuric and nitric acid. Primarily used as an industrial catalyst and in jewelry, palladium is a rare silver-white transition metal similar to platinum.
Obtained from: platinum, nickel, copper, mercury ores
